North America : Leading Innovation and Demand
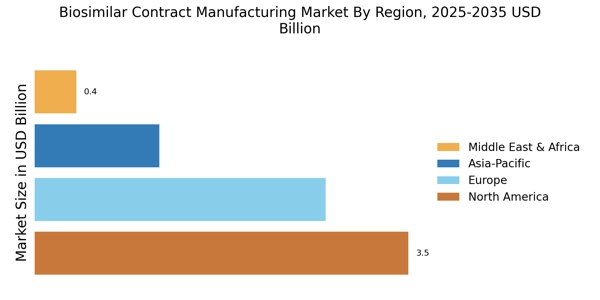
North America leads in the Biosimilar Contract Manufacturing Market size, accounting for over 45% of the global revenue in 2024. The region benefits from advanced healthcare infrastructure, increasing adoption of biosimilars, and supportive regulatory frameworks. The demand is driven by rising healthcare costs and the need for affordable biologics, with the FDA actively promoting biosimilar approvals to enhance competition and patient access.
- According to CDC, chronic diseases account for 90% of annual healthcare spending in the U.S., exceeding $4.1 trillion, while biologics represent a significant share of treatment costs, encouraging biosimilar adoption and boosting demand for contract manufacturing to improve affordability and accessibility.
The United States is the primary player in this market, with significant contributions from companies like Amgen and Catalent. Canada also plays a role, albeit smaller, in the biosimilars landscape. The competitive environment is characterized by the presence of major players such as Samsung Biologics and Lonza Group, which are expanding their manufacturing capabilities to meet the growing demand for biosimilars in the region.
Europe : Regulatory Support and Growth
Europe Biosimilar Contract Manufacturing Market size was valued at USD 2.32 billion in 2024, making it the second-largest regional market with a 30% share. The region's growth is fueled by a robust regulatory framework that encourages the development and approval of biosimilars, alongside increasing healthcare costs and a rising demand for affordable treatment options. The European Medicines Agency (EMA) has been instrumental in streamlining the approval process, which has led to a surge in biosimilar products entering the market.
- According to ECDC and EU data, Europe has approved over 80 biosimilars, while biosimilars have generated savings exceeding €30 billion, significantly improving healthcare sustainability and encouraging contract manufacturing growth to meet rising demand for affordable biologic therapies.
Leading countries in this region include Germany, the UK, and France, which are home to several key players like Boehringer Ingelheim and Sandoz. The competitive landscape is marked by a mix of established pharmaceutical companies and emerging biotech firms, all vying for a share of the growing biosimilars market. The presence of strong manufacturing capabilities and innovation in bioprocessing technologies further enhances Europe's position in the global biosimilar landscape.
Asia-Pacific : Emerging Market Potential
Asia-Pacific is rapidly emerging as a significant player in the biosimilar contract manufacturing market, holding approximately 15% of the global market share. The region's growth is driven by increasing healthcare expenditures, a large patient population, and a growing acceptance of biosimilars. Countries like China and India are leading this trend, supported by favorable government policies aimed at boosting local manufacturing and reducing healthcare costs.
China is the largest market in the region, with companies like Wuxi Biologics and Fujifilm Diosynth Biotechnologies leading the charge. India is also making strides with its robust pharmaceutical sector, focusing on biosimilars to cater to both domestic and international markets. The competitive landscape is evolving, with both local and international players investing in advanced manufacturing technologies to enhance production efficiency and quality.
Middle East and Africa : Untapped Market Opportunities
The Middle East and Africa region is gradually developing its biosimilar contract manufacturing market, currently holding about 5% of the global market share. The growth is primarily driven by increasing healthcare needs, rising awareness of biosimilars, and government initiatives aimed at improving healthcare access. Countries like South Africa and the UAE are at the forefront, with efforts to establish local manufacturing capabilities and attract foreign investment in the biosimilars sector.
The competitive landscape is still in its infancy, with a few local players and international companies exploring opportunities in the region. The presence of key players is limited, but there is significant potential for growth as governments focus on enhancing healthcare infrastructure and regulatory frameworks. As the market matures, it is expected to attract more investment and innovation in biosimilar manufacturing.